The original one was modified in order to suit to the current question in hand. Note: The galvanic cell is courtesy of ELI JONES. The salt bridge, then does his duty by neutralizing these two aqueous solutions (see the diagram). Your two half cells are $\ce$ deposits on cathode, and thus, the aqueous solution loses the silver cations, giving it a net negative charge. Now, let's see what chemical reactions are happening in your voltaic cell. The external circuit is used to conduct the flow of electrons between electrodes, which usually go through a load ( e.g., a light bulb or voltmeter) to close the circuit (see the diagram). The salt bridge connecting half-cells is a chamber of electrolytes, which are needed to maintain the neutrality of the solutions in both half-cells. The oxidation and reduction reactions are separated into compartments, which are called half-cells (each half-cell consists of electrode and the appropriate electrolyte solution). The anode is an electrode where oxidation occurs and the cathode is an electrode where reduction occurs. An electrochemical cell consists of two electrodes (anode and cathode), two electlolyte solutions, and a salt bridge: Instead, they migrate to the solution of the solution of the oxidation half cell.Ī voltaic cell is an electrochemical cell that uses a chemical reaction to produce electrical energy. They don't migrate to any electrode (neither cathode nor anode). Where do negative ions migrate from the salt bridge? Red cat: reduction occurs at the cathode. 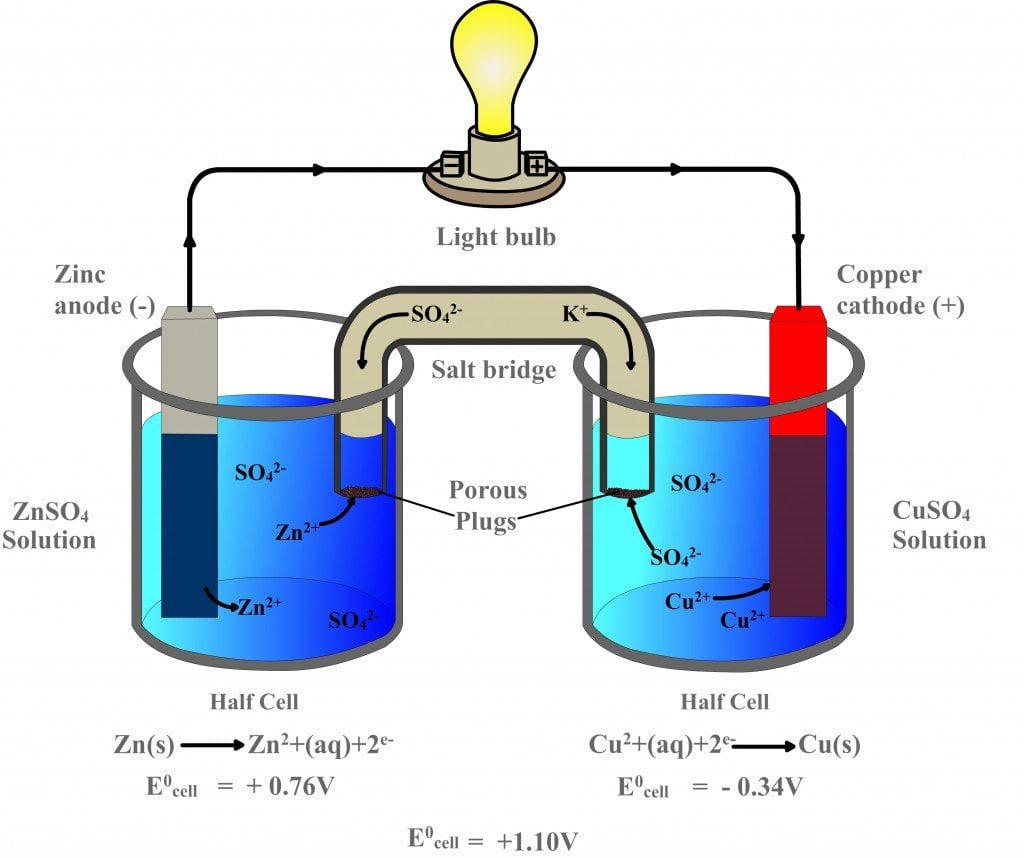
The cathode is the electrode at which electrons are accepted or consumed. Oil rig: oxidation is loss, reduction is gain (of electrons) The electrochemical cells have a cathode and an anode. The salt bridge provides anions (negative charged particles) to the solution of the oxidation half cell, and cations (positively charged particles) to the solution of the reduction half reaction. Basically, a salt bridge helps in migration of ions in oxidation-reduction half reactions occurring at anode and cathode. The electrons are supplied via the wire, and the silver cations leave the aqueous solution to be deposited on the cathode, giving the solution a net negative charge. A salt bridge is a device which helps in maintaining electrical neutrality between the solution at both ends of cathode and anode. Reduction half reaction (happens at the cathode) The electrons get removed via the wire, and the magnesium cations enter the aqueous solution, giving it a net positive charge. Accordingly, electrochemical cells used in batteries usually don't have a salt bridge but a semipermeable membrane instead, or a clever choice of half reactions where mixing of the electrolytes is not a problem (lead acid battery for example, where the only soluble species, sulfuric acid, is common to both half reactions).Oxidation half reaction (happens at the anode) This tells you that the salt bridge will be depleted after a certain amount of current flows through the wire. What causes the initial 2e- to move through the wire from the anode to the cathode I understand the entire process once. Once the Zn ion solution becomes too positive the salt bridge allows negative Sulfate ions to flow into the zinc side. To conclude, ions flow out of the salt bridge into the solutions A and B, but virtually no ion flows all the way through the salt bridge. Our battery stops working A salt bridge however balances the charges of the solutions on both sides. The solution in the salt bridge is not mixed, so it would take a very long time for the electrolyte in A to diffuse all the way to B. When you have a high concentration of inert ions in the salt bridge, cations in the salt bridge will flow into B, and anions in the salt bridge will flow into A. But Mg2 is NOT reduced, at least not in water environment. The idea of the salt bridge is to prevent electrolytes mixing while providing ion flow. 1 Mg is the cathode, you were already told Mg2 is not. According to my notes and many sources on the internet, electrons and cations both travel from the anode (A in the image) to the cathode (B in the image). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
